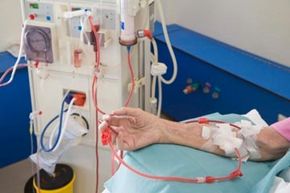
Imagine that you have multiple sclerosis (MS). This autoimmune disease causes your body's immune system to attack healthy cells, eventually leading you to lose control of the muscles in your limbs. Besides exercise and physical therapy to keep muscles as strong as possible, you don't have many options for treatment; however, a plasmapheresis procedure may offer hope for people suffering from MS and other autoimmune disorders.
Autoimmune diseases cause the body's immune system to attack itself. While some cells attack the immune system directly, other cells produce auto-antibodies that circulate through the blood to attack healthy cells. People diagnosed with autoimmune disorders usually take medication to suppress their immune systems or lessen the inflammation of tissues; however, use of these medications for long periods of time can have serious side effects. For example, medications can suppress the immune system so much that the body can't fight off infection.
Advertisement
In the 1970s medical researchers explored alternative treatments for autoimmune disorders that could help reduce the amount of medication patients had to take. One method that researchers discovered was plasmapheresis.
When a patient undergoes plasmapheresis procedure, plasma, the fluid part of the blood, is removed from blood cells by a cell separator. The separator spins blood at a rapid speed to separate blood cells from the plasma, or it passes the blood through a membrane with pores so tiny that only plasma can fit through. Cells are put back in the body while the plasma is disposed of and replaced with other liquids such as a saline and albumin combination, fresh frozen plasma, or a plasma substitute [source: Myasthenia Gravis Foundation of America] In a method similar to kidney dialysis treatment, this procedure removes auto-antibodies, contained in the plasma, from blood [source: Muscular Dystrophy Association].
Plasmapheresis procedures have been proven effective as treatment for autoimmune diseases that affect the limbs such as Lambert-Eaton syndrome, myasthenia gravis, chronic demyelinating polyneuropathy and Guillain-Barré syndrome. Its use as treatment for multiple sclerosis, polymyositis and dermatomyositis has not been determined [source: Muscular Dystrophy Association]. In addition to autoimmune disorders, plasmapheresis procedures can be used to treat patients with neurological disorders, blood toxins and high levels of cholesterol that can’t be controlled by medicine or diet [source: de la Rocha].
On the next page, we'll take a look at the experience of a patient during a plasmapheresis procedure and the complications to look out for if you're undergoing the treatment.
Advertisement